You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
US Navy Deep Submergence Rescue Vehicle (DSRV) Concepts
- Thread starter RyanC
- Start date
- Joined
- 9 October 2009
- Messages
- 21,969
- Reaction score
- 13,616
Deep Submergence Rescue Vessel Corrosion Studies
These studies were initiated (1) to determine the behavior of galvanic
couples composed of different alloy fasteners in hull materials, other dissimilar
couples, and welded alloys, and (2) the protective value of galvanic
anodes, paints and sealing compounds in seawater.
One set of specimens was removed after 6 months of exposure at the
surface in the Pacific Ocean and another set was removed after 1 year.
Sacrificial anodes protected aluminum alloys and type 321 stainless
steel from corroding; however, they did not afford complete protection to
the contacting surfaces of a titanium-aluminum galvanic couple. Four of
seven sealing compounds were satisfactory sealants at contacting surfaces.
Paint coatings prevented galvanic corrosion ot an aluminum alloy when in
contact with 6A1-4V titanium alloy and A-286 stainless steel only as long as
the paint coatings remained intact. After 6 months of exposure, there were
paint failures on seven of nine specimens and on 10 of 10 specimens after 1
year of exposure.
Effects of Environment on Coatings
A study was completed on the effects of pressures equivalent to a
depth of 6,000 feet in the ocean on the blistering of paints applied over
steel. It was found that blistering of paint in the deep ocean is similar to that
in the shallow ocean.
Steel specimens were coated with zinc-rich epoxies and a zinc-rich
Saran, with and without topcoats, and exposed in the deep ocean on STU 1-5.
After 6 months of exposure, the non topcoated zinc-rich organic coatings
were giving excellent protection to the steel panels, even along a scribed cut
made in the paints prior to exposure. Of the test specimens below, the lowerleft
panel was not topcoated and shows bright steel at the scribe maik. However,
when the panels were topcoated with a catalyzed epoxy (lower right),
a Navy alkyd (uipper left), and a silicone alkyd over the Navy alkyd (upper
right), varying degrees of corrosion occurred.
OCEAN ENGINEERING PROGRAM, MARCH 1968-MARCH 1969.
- Joined
- 9 October 2009
- Messages
- 21,969
- Reaction score
- 13,616
http://www.dtic.mil/dtic/tr/fulltext/u2/480384.pdf
http://www.dtic.mil/dtic/tr/fulltext/u2/679230.pdf
http://www.dtic.mil/dtic/tr/fulltext/u2/854434.pdf
http://www.dtic.mil/dtic/tr/fulltext/u2/a005879.pdf
http://www.dtic.mil/dtic/tr/fulltext/u2/679230.pdf
http://www.dtic.mil/dtic/tr/fulltext/u2/854434.pdf
http://www.dtic.mil/dtic/tr/fulltext/u2/a005879.pdf
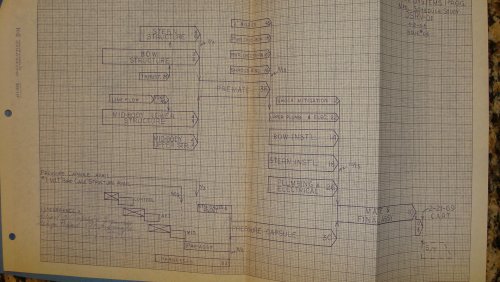
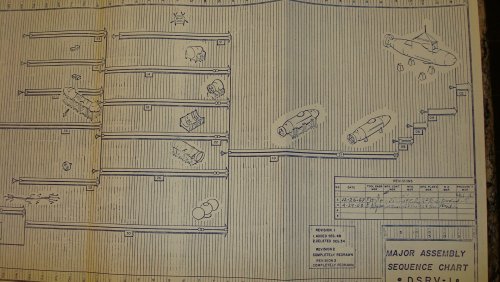
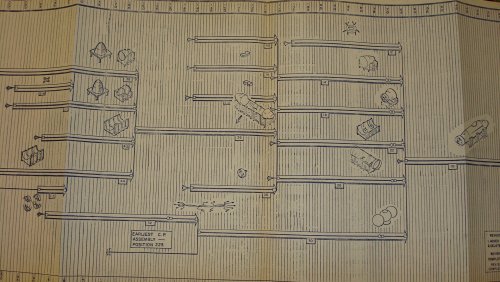
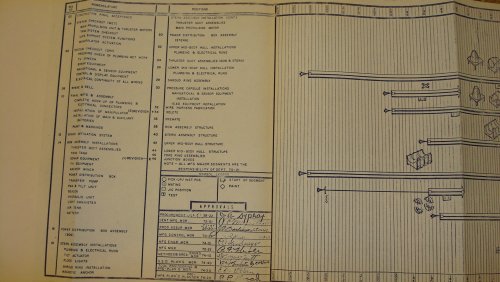
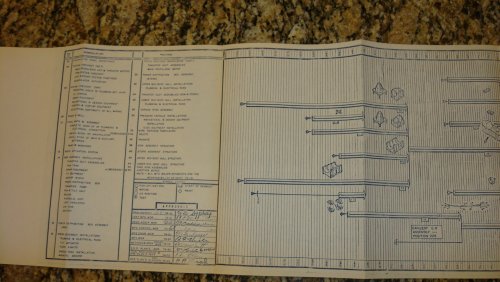
I AM POSTING THIS TONIGHT ON EBAY ..... ITS A MANUFACTURING PROGRAM PLAN #N606-5, LOCKHEED MODEL DSRV-1 ...... BY THE NASA/DSRV PROGRAMS .....ENJOY ..
Attachments
- Joined
- 9 October 2009
- Messages
- 21,969
- Reaction score
- 13,616
jmnjohn said:I AM POSTING THIS TONIGHT ON EBAY ..... ITS A MANUFACTURING PROGRAM PLAN #N606-5, LOCKHEED MODEL DSRV-1 ...... BY THE NASA/DSRV PROGRAMS .....ENJOY ..
Perhaps John's post should be moved or copied over to this topic in Naval Projects?
- Joined
- 5 May 2007
- Messages
- 351
- Reaction score
- 408
Recently posted by Periscope Films on YouTube, a 1966(?) film detailing part of North American's DSRV efforts, entitled "North American Aviation's (NAA) Deep Submergence Systems Program Office Deep Submergence Rescue Vehicle Mockup Studies."
Periscope Films describes it accordingly:
"Made in 1966 as part of North American Aviation's bid to produce the U.S. Navy's Mystic Class DSRV Deep Submergence Rescue Vehicle, this rare and unique film gives a behind-the-scenes look at the development of this revolutionary rescue submersible. The film shows how aircraft and spacecraft simulators were used to study the problems that the DSRV would encounter, and two different mock-ups were designed and built to demonstrate (1) a spherical hull design and (2) a cylindrical hull design. . . ."
The title card at the very beginning of the film states:
"This film is Volume VIII Appendix 18 of the North American Aviation, Inc. proposal responsive to the US Navy RFP No. SP001-0139-66 for a Deep Submergence Rescue Vehicle."
Periscope Films describes it accordingly:
"Made in 1966 as part of North American Aviation's bid to produce the U.S. Navy's Mystic Class DSRV Deep Submergence Rescue Vehicle, this rare and unique film gives a behind-the-scenes look at the development of this revolutionary rescue submersible. The film shows how aircraft and spacecraft simulators were used to study the problems that the DSRV would encounter, and two different mock-ups were designed and built to demonstrate (1) a spherical hull design and (2) a cylindrical hull design. . . ."
The title card at the very beginning of the film states:
"This film is Volume VIII Appendix 18 of the North American Aviation, Inc. proposal responsive to the US Navy RFP No. SP001-0139-66 for a Deep Submergence Rescue Vehicle."
Last edited:
- Joined
- 5 May 2007
- Messages
- 1,482
- Reaction score
- 2,858
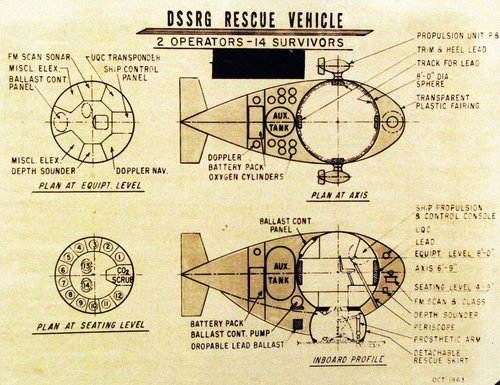
The original concept for the DSRV, as proposed by the Deep Submergence System Review Group in 1963.
 www.history.navy.mil
www.history.navy.mil
Should a submarine sink, a proposed rescue vehicle which will carry to operators and 14 survivors, shown in this diagram, is recommended by the Deep Submergence Systems Review Group. Master Caption: Following the loss of the submarine USS Thresher in April 1963, the Secretary of the Navy established the Deep Submergence Systems Review Group to analyze the requirements for placing man at great ocean depths. The study included an examination of plans for the development and procurement of components and systems related to the location, identification, rescue from and the recovery of large and small boats, including submarines, from the ocean floor.
330-PSA-99-64 (USN 711460): Undersea Recovery Vehicle
Undersea Recovery Vehicle. Should a submarine sink, a proposed rescue vehicle which will carry to operators and 14 survivors, shown in this diagram, is recommended by the Deep Submergence Systems Review Group. Master Caption: Following the loss of the submarine USS Thresher in April 1963, the...
Attachments
- Joined
- 9 October 2009
- Messages
- 21,969
- Reaction score
- 13,616
Scott Kenny
ACCESS: USAP
- Joined
- 15 May 2023
- Messages
- 11,628
- Reaction score
- 14,304
It's really too bad the US retired the DSRVs without direct replacement. Yes, we've still got McCann Rescue Chambers, but those depend on a submarine being more or less upright on the bottom.
A DSRV had some fancy mercury ballast tanks to allow it to roll up to 45deg to seal itself onto a sub in trouble. Not that such a thing was ever tested, to my knowledge.
A DSRV had some fancy mercury ballast tanks to allow it to roll up to 45deg to seal itself onto a sub in trouble. Not that such a thing was ever tested, to my knowledge.
bobthemonkey
ACCESS: Restricted
- Joined
- 9 February 2011
- Messages
- 31
- Reaction score
- 89
The SRDRS is rated to a 45 degree mating angle with DISSUB. https://www.navy.mil/Resources/Fact...e/2169620/undersea-rescue-command-urc-assets/It's really too bad the US retired the DSRVs without direct replacement. Yes, we've still got McCann Rescue Chambers, but those depend on a submarine being more or less upright on the bottom.
A DSRV had some fancy mercury ballast tanks to allow it to roll up to 45deg to seal itself onto a sub in trouble. Not that such a thing was ever tested, to my knowledge.
Obviously this is a tethered ROV-style setup, as opposed to a full free floating option like the DSRVs.
- Joined
- 5 May 2007
- Messages
- 1,482
- Reaction score
- 2,858
On the other hand, it can do transfer under pressure, which was always a major shortcoming of the DSRV system.Obviously this is a tethered ROV-style setup, as opposed to a full free floating option like the DSRVs.
Scott Kenny
ACCESS: USAP
- Joined
- 15 May 2023
- Messages
- 11,628
- Reaction score
- 14,304
If a sub is letting water into the hull at depth, the available air volume is minimal. I'll keep myself out of jail and stick with the "In excess of 800 feet" numbers officially published.On the other hand, it can do transfer under pressure, which was always a major shortcoming of the DSRV system.
At 800ft down, the external pressure is some 350psi. 24bar, if you think in that better. And honestly, well, it's easier to do this one in bar of pressure, not psi. A sub at 800ft down that has flooded to equilibrium pressure will only have 1/24 the hull volume still dry.
- Joined
- 5 May 2007
- Messages
- 1,482
- Reaction score
- 2,858
You don't even need to get that deep, or flood to equilibrium pressure, for TUP to be needed. Just losing a couple of compartments and burning oxygen candles will bring the survivors up to a couple of atmospheres of pressure. IIRC 4 bar was expected before a rescue craft could be on top.A sub at 800ft down that has flooded to equilibrium pressure will only have 1/24 the hull volume still dry.
Because of the inevitable delay in getting the rescue craft there, the survivors will become saturated, meaning any rescue needs to consider decompression sickness.
When the SRDRS program started, certain US boats (I think some of the 637s) were equipped to pressurise the entire boat, but not all internal equipment was rated to allow the operation. The only TUP capability in NATO was the VANGUARD class submarines, which were designed to allow the forward part of the boat to be pressurised to (IIRC) 2.8 bar. But that means you need the UK to weaken its deterrent to go rescue your submariners.
SRDRS was originally envisaged as operating alongside the DSRVs, rather than replacing them: ROVs to clear debris and pass supplies, and a deck decompression chamber for survivors brought up by the DSRV.
Scott Kenny
ACCESS: USAP
- Joined
- 15 May 2023
- Messages
- 11,628
- Reaction score
- 14,304
You seem to be under a misconception as to how many watertight compartments a sub has. A US boat only has 3 or 4, and only 2 or 3 are habitable. An Ohio class has 4 watertight compartments: Forward/ops compartment, missile compartment, reactor compartment, and engineroom. A 688 has 3 compartments: forward/ops compartment, reactor compartment, and engineroom. RC is not habitable in either case. If one of those compartments flooded, you'd better pray it was forward compartment.You don't even need to get that deep, or flood to equilibrium pressure, for TUP to be needed. Just losing a couple of compartments and burning oxygen candles will bring the survivors up to a couple of atmospheres of pressure. IIRC 4 bar was expected before a rescue craft could be on top.
Because of the inevitable delay in getting the rescue craft there, the survivors will become saturated, meaning any rescue needs to consider decompression sickness.
When the SRDRS program started, certain US boats (I think some of the 637s) were equipped to pressurise the entire boat, but not all internal equipment was rated to allow the operation. The only TUP capability in NATO was the VANGUARD class submarines, which were designed to allow the forward part of the boat to be pressurised to (IIRC) 2.8 bar. But that means you need the UK to weaken its deterrent to go rescue your submariners.
SRDRS was originally envisaged as operating alongside the DSRVs, rather than replacing them: ROVs to clear debris and pass supplies, and a deck decompression chamber for survivors brought up by the DSRV.
Ohios also had "salvage air" valves in the piping, so could be operated at greater than 1atm. I believe that's standard in other US boats as well. I would not WANT to do that, but it could be done.
The big problem is the lack of medical personnel on submarines. I'd assume that you'd need to grab one of the SSGNs, and transfer off a quarter or more of their crew (curated so that the remaining crew can still safely operate the ship for the 6 weeks decompression is going to take). Then you'd need to take on a dozen or so doctors, nurses, and corpsmen to assist in caring for the injured. Because the typical USN sub crew only has a single Independent Duty Corpsman, not even a full doctor.
- Joined
- 5 May 2007
- Messages
- 1,482
- Reaction score
- 2,858
Probably thinking of Soviet/Russian boats, which do have more (still not many) watertight bulkheads, but really the point is that you don't need to flood the entire pressure hull. All you need is some kind of accident where you can't surface and can't control the atmosphere, and eventually pressure will build up.You seem to be under a misconception as to how many watertight compartments a sub has.
Unfortunately I can't find the blasted report... but it was an official study, published either by the USN or NATO around 1990, which flagged these issues.The big problem is the lack of medical personnel on submarines. I'd assume that you'd need to grab one of the SSGNs, and transfer off a quarter or more of their crew (curated so that the remaining crew can still safely operate the ship for the 6 weeks decompression is going to take). Then you'd need to take on a dozen or so doctors, nurses, and corpsmen to assist in caring for the injured. Because the typical USN sub crew only has a single Independent Duty Corpsman, not even a full doctor.
The ability of some USN boats to raise the internal pressure was noted. At that time it wasn't universal, and there were concerns that internal equipment couldn't survive the higher pressure for more than a limited amount of time. The VANGUARD class were explicitly noted as being able to operate the forward compartment at higher pressure, but that there was insufficient space to accommodate a full complement of survivors.
The difficulties associated with crewing were also noted. Not just in terms of bringing aboard sufficient medical personnel while being able to operate the boat, but also finding somewhere to put the survivors. Presumably the entire torpedo compartment would be turned into additional berthing as a minimum.
It's worth noting that the design of NSRS and SRDRS assumes that decompression times will be measured in hours, rather than days or weeks - i.e. that a disabled submarine wouldn't be flooded to equilibrium at anything approaching maximum operating depth.
Vepr157
I like submarines
- Joined
- 20 December 2015
- Messages
- 47
- Reaction score
- 66
@Yellow Palace, is this the report you're thinking of? A SHIPALT was carried out to some 637s which allowed the forward compartment to be pressurized to several bar to serve as a decompression chamber.
- Joined
- 5 May 2007
- Messages
- 1,482
- Reaction score
- 2,858
Yes, that's what I was thinking of. Some of the details have evidently faded in my memory - the USN's inability to carry out TUP was procedural, rather than technical.@Yellow Palace, is this the report you're thinking of? A SHIPALT was carried out to some 637s which allowed the forward compartment to be pressurized to several bar to serve as a decompression chamber.
Scott Kenny
ACCESS: USAP
- Joined
- 15 May 2023
- Messages
- 11,628
- Reaction score
- 14,304
Well, there's certainly not a lot of shallow water where you could recover a downed sub crew from. Most of the Pacific islands have the 100-fathom curve about 1nmi off the shoreline. If you can get your sub to a point where the crew could conceivably be rescued, couldn't you just run the thing aground a little closer to the shore?It's worth noting that the design of NSRS and SRDRS assumes that decompression times will be measured in hours, rather than days or weeks - i.e. that a disabled submarine wouldn't be flooded to equilibrium at anything approaching maximum operating depth.
But I think the decompression time also includes how long someone was at pressure. And a disabled sub would be at increased pressure for days to a week before rescue gear could arrive on scene.
And Parche definitely didn't do some sketchy stuff involving long term saturation diving and decompression...@Yellow Palace, is this the report you're thinking of? A SHIPALT was carried out to some 637s which allowed the forward compartment to be pressurized to several bar to serve as a decompression chamber.
- Joined
- 5 May 2007
- Messages
- 1,482
- Reaction score
- 2,858
There are places it makes sense. But most of the Pacific, if you lose buoyancy the best case scenario is that you have just enough time to bend over and kiss your ass goodbye.Well, there's certainly not a lot of shallow water where you could recover a downed sub crew from. Most of the Pacific islands have the 100-fathom curve about 1nmi off the shoreline. If you can get your sub to a point where the crew could conceivably be rescued, couldn't you just run the thing aground a little closer to the shore?
John Craven's 2,000/20,000 foot rule for design depths makes a lot of sense when you start looking into it. The factors of safety applied to submarine pressure hulls are published. Any conclusions drawn about operating depths are your own.
Yup, the survivors would be saturated at whatever the ambient pressure inside the pressure hull was unless they could escape quickly. No more than 5 bar for the DSRV to be able to operate, with decompression in the DSRV to between 2 and 4 bar for the MOSUB, or to atmospheric if no TUP was available – that would limit the DISSUB internal pressure to about 1.8 bar.And a disabled sub would be at increased pressure for days to a week before rescue gear could arrive on scene.
The design of the NSRS decompression chambers limits the number of rescuees under decompression to 72 at a time. That implies that the first batch of 18 will be done in the time it takes the fifth batch to be rescued. I'm not 100% sure what SRDRS can handle, but suspect it's similar.
Of course not. I'm shocked that you could imply such a thing.And Parche definitely didn't do some sketchy stuff involving long term saturation diving and decompression...
I've talked before about the difference between 'deploying' and 'non-deploying' navies. Submarine rescue is one of those areas. A deploying navy could need to rescue its submariners anywhere in the world, so a flyaway system makes a lot of sense. The 24 hours to mobilise is small compared to the time needed to get a dedicated rescue ship to the scene. For a non-deploying navy, the converse is true. A lost submarine is likely to be relatively close to home, so having the kit loaded onto a dedicated ship and ready to go makes a lot of sense.
Of course, there aren't many non-deploying navies that have the budget for their own submarine rescue capability. But looking at who has submarine rescue ships and who has flyaway systems is a bit of a clue as to which navies are which. The Soviet Union started building some really big, really fancy submarine rescue ships.
What's the point? Well, the USN is clearly a deploying navy. But it built two submarine rescue ships (ORTOLAN and PIGEON) that didn't have TUP capability. And which couldn't get to a DISSUB in time to do anything useful, even if anything useful was capable of being done. But they did have an elaborate saturation diving system, and an equally elaborate launch and recovery system for submersibles.
I know at least one mission that they were intended for, but none of the rest of the hardware (bar one prototype) was constructed. I suspect other missions were envisaged, and some of them may have been carried out, but I don't know what they were.
Scott Kenny
ACCESS: USAP
- Joined
- 15 May 2023
- Messages
- 11,628
- Reaction score
- 14,304
Yeah, the TRE inspectors didn't like my attitude when I reminded them of that fact in 2002 or so. They'd just started pushing submarine escape stuff at that time.most of the Pacific, if you lose buoyancy the best case scenario is that you have just enough time to bend over and kiss your ass goodbye.
Got a link? I don't think I've seen that, and now I'm curious.John Craven's 2,000/20,000 foot rule for design depths makes a lot of sense when you start looking into it. The factors of safety applied to submarine pressure hulls are published. Any conclusions drawn about operating depths are your own.
- Joined
- 5 May 2007
- Messages
- 1,482
- Reaction score
- 2,858
It's in his book The Silent War. The idea is that there are only really two depths that make sense as limits: 2,000 feet lets you access anywhere on the continental shelf, and 20,000 feet lets you access anywhere on the abyssal plain except the deepest ocean trenches. Therefore anything which is going to be exposed to ocean pressure should be designed for one of those two depths.Got a link? I don't think I've seen that, and now I'm curious.
Where it gets interesting is the AUTEC submersibles (designed for 6,500 feet) and the DSRVs (designed for 6,000 feet, but not achieved because welding HY-130 is hard). That's too deep for the continental shelf, and not deep enough for the abyssal plain. But plenty deep enough to get down to the SOFAR channel....
Scott Kenny
ACCESS: USAP
- Joined
- 15 May 2023
- Messages
- 11,628
- Reaction score
- 14,304
I'm going to quibble because that doesn't get into subs that might want to get down into the SOFAR channel for tactical reasons. Say, 3500ft test depth.It's in his book The Silent War. The idea is that there are only really two depths that make sense as limits: 2,000 feet lets you access anywhere on the continental shelf, and 20,000 feet lets you access anywhere on the abyssal plain except the deepest ocean trenches. Therefore anything which is going to be exposed to ocean pressure should be designed for one of those two depths.
And because designing for 20,000ft down means battleship thick hulls or worse.
Well, of course the Atlantic Undersea Test and Evaluation Center needs access to the SOFAR channel. And I think the DSRVs were intended to get that deep to reach any sub that sunk on the continental shelf including any of the canyons.Where it gets interesting is the AUTEC submersibles (designed for 6,500 feet) and the DSRVs (designed for 6,000 feet, but not achieved because welding HY-130 is hard). That's too deep for the continental shelf, and not deep enough for the abyssal plain. But plenty deep enough to get down to the SOFAR channel....
- Joined
- 5 May 2007
- Messages
- 1,482
- Reaction score
- 2,858
He was mostly concerned with moderately sized (say NR-1 at the largest) submersibles doing stuff on the seabed. Submarines as such were a useful way of not having to interact with the surface, but the seabed was where you wanted to be.I'm going to quibble because that doesn't get into subs that might want to get down into the SOFAR channel for tactical reasons. Say, 3500ft test depth.
Part of the thinking was that it's much easier to build a new pressure hull than to build a new gadget to go outside it. So design your manipulators, motors etc for the maximum reasonable depth and you've got future proofing.And because designing for 20,000ft down means battleship thick hulls or worse.
Thing is, there needs to be something left to get to. And that means either a very deep diving submarine, or something that isn't a very deep diving submarine but which would still need crew transfer. NR-1 operated at the depths concerned, but you couldn't mate to it. There's nothing else in the West that fits the bill.And I think the DSRVs were intended to get that deep to reach any sub that sunk on the continental shelf including any of the canyons.
Future proofing? Maybe.
Scott Kenny
ACCESS: USAP
- Joined
- 15 May 2023
- Messages
- 11,628
- Reaction score
- 14,304
Definitely for research and "research" ie "seeing what the enemy lost".He was mostly concerned with moderately sized (say NR-1 at the largest) submersibles doing stuff on the seabed. Submarines as such were a useful way of not having to interact with the surface, but the seabed was where you wanted to be.
Not sure I agree with that, but it's been a long time since he wrote that idea.Part of the thinking was that it's much easier to build a new pressure hull than to build a new gadget to go outside it. So design your manipulators, motors etc for the maximum reasonable depth and you've got future proofing.
Dolphin could go very deep, and Thresher imploded at something like double her test depth.Thing is, there needs to be something left to get to. And that means either a very deep diving submarine, or something that isn't a very deep diving submarine but which would still need crew transfer. NR-1 operated at the depths concerned, but you couldn't mate to it. There's nothing else in the West that fits the bill.
Similar threads
-
-
-
-
-
The United States Navy's "Rock-Site" concept for underwater base
- Started by Michel Van
- Replies: 7